Discover the ground-breaking advancements of WS23, a cooling agent that is reshaping sensory experiences across various industries with its cutting-edge technology.
2-Isopropyl- N,2,3-trimethylbutanamide, otherwise called WS23, finds versatile applications in candies, beverages, oral care, skin care products, and pharmaceuticals. Its enhanced cooling properties and compatibility with diverse flavours elevate sensory experiences and address challenges in taste-centric product development.
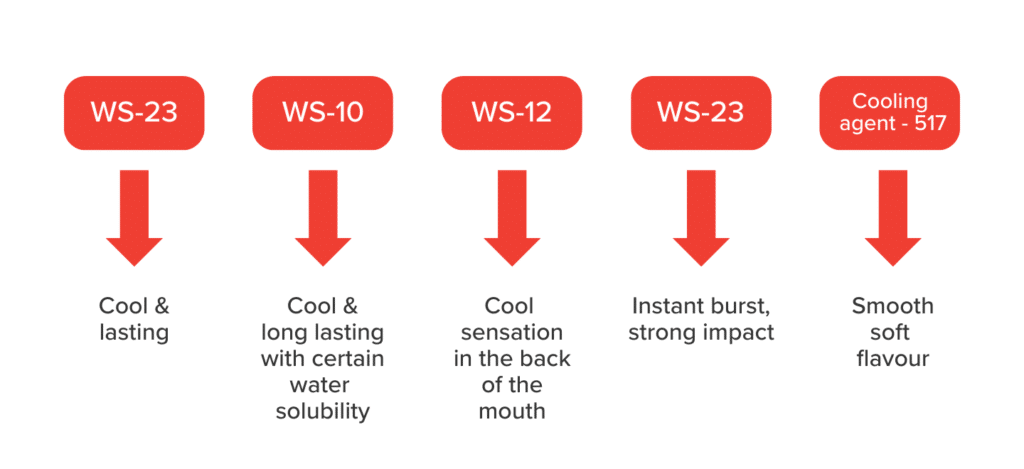
WS23 is a prominent member of an esteemed series of cooling agents. Its market leadership is reinforced by solid demand from renowned brands like P&G, Mondelez, and Mars in the U.S.
The WS series of cooling agents offers a variety of options, including WS-1, WS-3, WS-10, WS-12, and WS-27, each with varying levels of cooling potency and duration. Among these, WS-3 and WS-23 are the most favoured choices.
Key Features
- Innovative Cooling Sensation: A distinctive and enduring cooling effect lasting 15 to 30 minutes. WS23 sets itself apart from the discomfort often associated with cooling agents derived from menthol or peppermint.
- Uncompromised Thermal Stability: Designed to withstand temperatures up to 200°C, it is an ideal choice for high-temperature applications such as baking and heat-based industries.
- Flavour Compatibility: WS23 seamlessly blends with various flavours, making it an adaptable addition to different product categories.
- Efficiency Optimization: Achieve a potent cooling effect with a minimal dosage range of 30-100mg/kg, leading to substantial cost benefits for manufacturers.
Technical Specifications
This white crystalline solid powder exhibits a distinctive mint-like cool aroma that sets it apart. Notably, WS23 boasts a melting point of 60-63°C and a solubility profile that renders it slightly soluble in water while finding compatibility with alcohol, Propylene Glycol, and various oils as efficient solvents.
With a content purity of no less than 99.0%, WS23 has garnered recognition from different authoritative bodies, including FEMA (3804), JECFA (1595), and Flavis (16.053). This compound’s multifaceted attributes open the door to various potential applications across industries, offering a refreshing avenue for altering sensory encounters.
How can we help?
We eagerly anticipate the ongoing success and innovation that WS23 Isopropyl Trimethyl Butyramide will bring to our portfolio and industries.
Contact your dedicated Redox representative and one of our team members will get back to you.
Protein supplements are gaining immense popularity worldwide, not just among professional athletes but also among a broader range of consumers who recognize their incredible benefits for sports nutrition, muscle building, and overall health. However, the traditional animal and plant-based protein options have limitations, necessitating the search for innovative alternatives. One such exciting product is yeast protein.
Protein has been shown to have numerous positive effects, including:
- Weight management,
- muscle building,
- tissue recovery,
- strength gains, and
- improved body composition.
These benefits have propelled the supplement market to a staggering value of $6.3 billion in 2021, with further growth projected in the coming years.
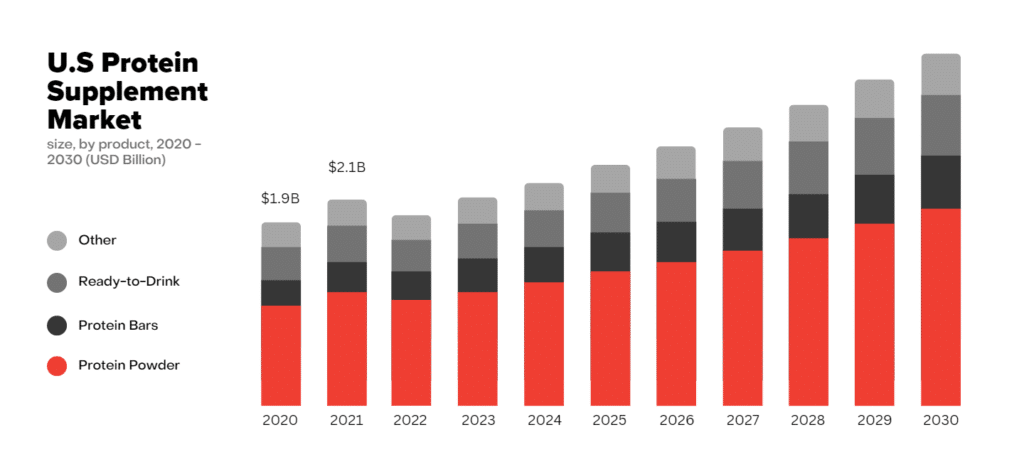
The global protein supplements market was valued at USD 6.26 billion in 2021 and is expected to expand at a compound annual growth rate (CAGR) of 8.0% from 2022 to 2030. The market is gaining momentum owing to the increasing number of health-conscious consumers and fitness centres around the globe.
The need for new protein sources
Our planet is facing a significant challenge in meeting the growing global demand for protein. Over the past two decades, protein consumption has been on the rise, and this trend is expected to continue as the population grows and economies thrive. However, relying solely on traditional animal agriculture to meet this demand poses a problem. In light of this concern, both companies and consumers are now turning their attention to yeast protein as a promising and sustainable solution.
Protein supplements come from various sources such as whey, casein, beef, fish, egg, soy, pea, hemp, and rice. Animal proteins, like whey, are often considered superior to plant-based options due to their rich content of essential amino acids. Moreover, plant-based proteins may not be as easily digested in the gut and can negatively affect metabolism and immune response.
Whey protein, derived from milk during cheese production, has long been regarded as the gold standard. It is a complete protein that is quickly digested and contains high levels of branched-chain amino acids (BCAAs) and leucine, which are crucial for muscle growth and recovery.
However, using animal sources in whey protein presents a significant barrier for many consumers. Environmental concerns, animal welfare issues, and contamination fears have prompted a global shift towards reducing animal product consumption. For instance, nearly half of the population in Singapore has already reduced or eliminated animal products from their diets. Similar trends are observed in other Asian countries, where many individuals opt for meat reduction. As a result, there is a growing demand for alternatives.
Challenges with plant-based proteins
Plant-based proteins have gained popularity as an alternative but still have limitations. They often lack the same amino acid content, digestibility, and protein synthesis capacity as animal proteins. Additionally, large-scale cultivation of plant-based protein sources, such as wheat, soy, and peas, requires substantial land resources and can be impacted by weather conditions. Concerns about heavy metals, pesticide residues, and genetically modified organisms further dampen consumer enthusiasm.
The Rise of the Alternative: Yeast Protein
Yeast proteins are an exciting and promising alternative. Nutritional yeast, commonly available in the market, is often known for its immune-boosting and gut health benefits. However, it is also a rich microbial protein source with high BCAA content.
While yeast protein has been studied for its nutritional value in animal agriculture for several decades, research on its impact on humans has been limited.
The properties and composition of AngeoPro
AngeoPro, derived from Saccharomyces cerevisiae, commonly known as brewer’s or baker’s yeast, contains over 70% high-quality protein and has a digestibility value similar to whey protein. It boasts a leucine content comparable to whey protein and even higher levels of total BCAAs. Acting as a “slow protein,” AngeoPro provides a steady and continuous supply of amino acids.
AngeoPro closely resembles animal proteins, outperforming low-quality plant proteins regarding nutritional value. Additionally, AngeoPro, as a fermentation product, offers advantages such as a reliable supply chain and reduced land requirements. The wastewater generated during AngeoPro production is repurposed as organic fertilizer for yeast cultivation, creating a more sustainable production process.
How Can Redox Help?
When it comes to meeting the surging demand for sustainable and high-quality protein, look no further than Redox as your trusted partner.
With yeast protein emerging as a revolutionary solution, Angel Yeast stands at the forefront of research and development, ensuring a future of protein supplementation that resonates with consumer preferences.
Contact us today to explore how Redox can be the essential catalyst in your sourcing strategy, providing you with unmatched expertise and top-notch solutions.
Produced from carbon-rich organic materials, activated carbon’s superior performance as an adsorbent lends itself to a growing list of applications as environmental regulations continue to become more stringent.
Activated carbon is a game-changing adsorbent, revolutionising how industries tackle contaminants and unwanted particles in liquids or gases. Pollution control efforts are particularly taking advantage of its capabilities, with reports pointing towards an optimistic future – all thanks to the growing call for environmental solutions globally.
How is Activated Carbon being used in industry?
Activated carbon is an incredibly versatile compound that serves a range of diverse functions.
- From the granular medium traditionally used to trap pollutants from liquids and gases to
- pellets designed explicitly for tackling volatile organic compounds (VOCs) and Hydrogen Sulfide (H2S) in gas filtration,
- it even comes in powder form – which can be added directly into liquid or soil to absorb toxins easily.
Its remarkable properties have enabled some incredible advancements across multiple industries.
While Activated carbon has become a powerful ally in multiple industries, from purifying potable and wastewater to cleaning out toxic gases from the air we breathe, it also plays an essential role in soil remediation by removing dangerous pollutants like PFAS and other organic compounds.
Even beverages can benefit– with Activated carbon able to strip wine and juices of their unwanted colour or toxins, such as patulin found commonly in apple juice!
Natures renewable resource
Customers are increasingly turning to sustainable solutions – unlike other forms of activated carbon, coconut shells and wood are renewable resources, each with unique properties when it comes to removing impurities.
In particular coconut shells are an incredibly sustainable solution to global raw material needs. Renewable and accessible within six months, coconuts outshine common alternative materials such as coal or wood that can take millions of years and decades to form. Not only do coconut trees capture CO2 from the atmosphere, but their harvesting is a by-product of our food industry – making them earth-friendly and beneficial in other ways.
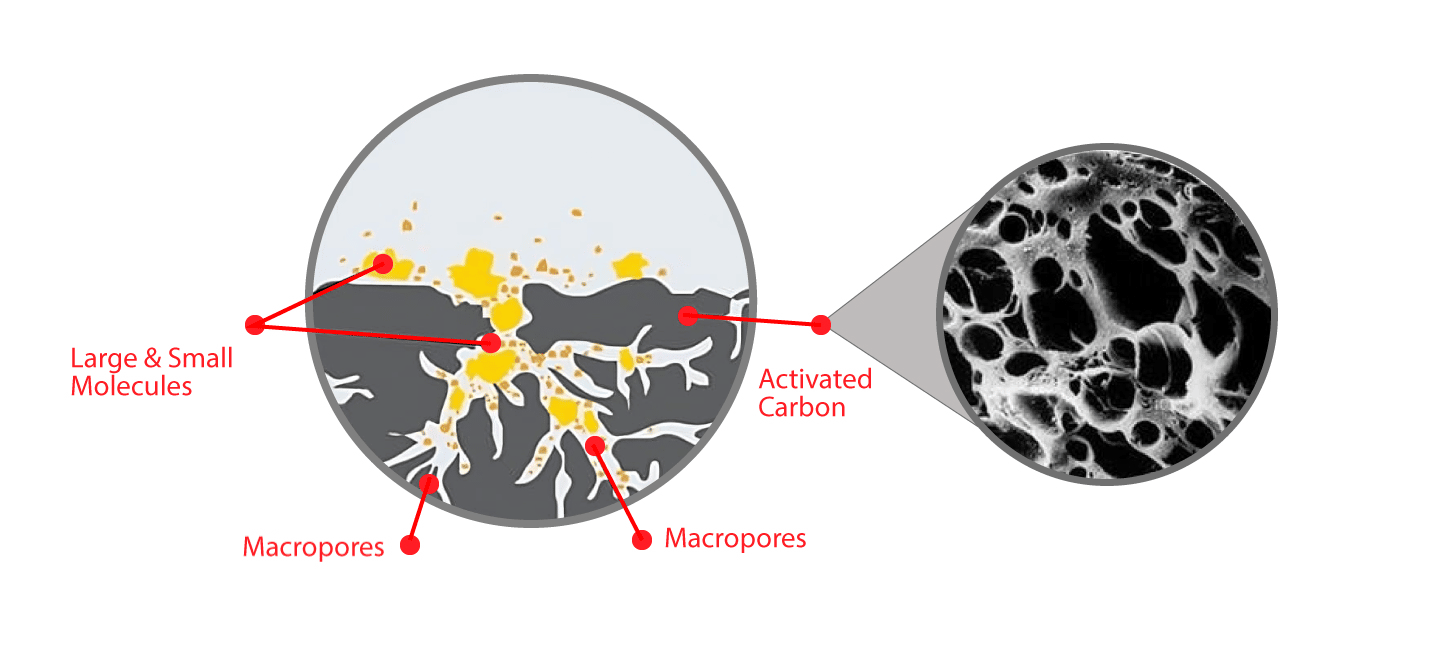
Activated carbon is an effective adsorbent because it is a highly porous material and provides a large surface area to which contaminants may adsorb.
Using it to produce activated carbon prevents it from being wasted and potentially burnt as fuel. In fact, every part of the coconut has a use, and there is no waste. The water, meat, husk and shell are all used across various industries.
Recent market reports point to an optimistic future driven by demand for its environmental uses. With a heightened awareness surrounding eco-friendly products like coconut-based activated carbon, new possibilities present daily as we uncover just how powerful this substance truly is in the fight against pollution.
How can we help?
Despite a range of competitor adsorbents available, activated carbon continues to lead the way across numerous applications. Thanks to solid growth prospects in key regions and industries, demand for this versatile material is set to remain healthy in the coming years. As a result, companies providing high-quality activated carbon will be well-positioned to capitalise on this trend.
Redox proudly offers a vast selection of activated carbon tailored to suit any application. From 25kg bags up to 500 kg bulker and with impregnation options – Redox only sources the highest quality carbons to suit your application through our trusted partner.
Unlock the potential of your sourcing strategy with Redox. Our experts are ready to help you realise a new level of success. Contact one of our experts today.
Not long after, French chemist Nicolas Leblanc produced sodium carbonate in 1791; pharmacist Valentin Rose the Younger discovered Sodium Bicarbonate in 1801. Neither Leblanc nor Younger could have imagined their discoveries’ impact on future generations.
How is it made, or where do we find it?
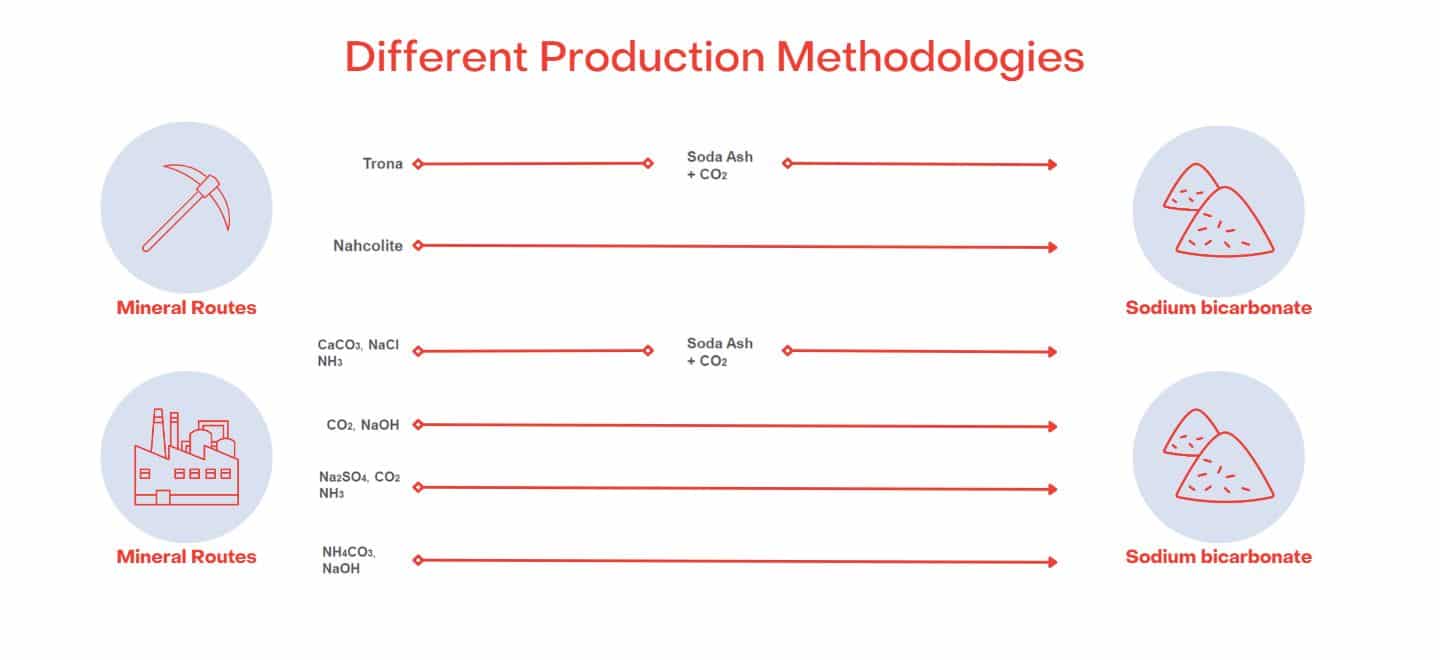
Sodium Bicarbonate is generally synthetically manufactured using the Solvay or the Trona Process.
The Solvay process is an industrial method used to produce Sodium Bicarbonate, commonly known as baking soda. The Solvay process is the reaction of Sodium Chloride, ammonia, and carbon dioxide in water.
Alternatively, the Trona process converts naturally formed/mined soda ash into bicarb.
However, it can also be found naturally occurring in evaporated lake basins worldwide. The largest commercially viable source is Natural Soda in Colorado, USA, which has full organic certification.
Different uses of Sodium Bicarbonate in the food & beverage industry
The flexibility of Sodium Bicarbonate is an undeniable asset for many industries, none more so than the food and beverage industry.
In baked goods, it acts as a leavening agent that produces carbon dioxide when combined with an acidic ingredient such as vinegar or lemon juice. This causes the dough to rise, creating lighter and fluffier baked goods like cakes and muffins. Not only that, Sodium Bicarbonate can also be used to make carbonated beverages like sodas and sparkling waters by dissolving it into water.
The dissolved sodium bicarbonate can then be added to other ingredients like flavourings, syrups, and colourings to create a refreshingly fizzy drink.
Lastly, Sodium Bicarbonate can also be used to tenderise meats for marinades or sauces, as its alkaline nature helps break down tough fibres in the meat’s muscles.

Sodium Bicarbonate or bicarbonate of soda, is a popular baking ingredient. It gives foods like bread, cakes, muffins, and cookies a light, fluffy texture.
Sodium Bicarbonate’s application in animal feed
Sodium bicarbonate is a vital ingredient in animal feed due to its numerous benefits.
It serves as an excellent source of electrolytes essential for healthy bodily functions. It also helps to maintain proper pH levels in the body, which improves digestion and increases nutrient uptake.
In addition, Sodium Bicarbonate can help reduce the build-up of lactic acid in the muscles, allowing animals to perform better during strenuous activity. Besides that, it can also act as a buffer against digestive upsets such as acidosis and scours and protect against microbial toxins.
All these benefits make Sodium Bicarbonate an essential ingredient in animal feed for optimum health and performance.
But its utility goes beyond the Food and Feed industries. For example:
- It’s commonly used in water treatment and swimming pools for pH adjustment.
- Being an alkali, it assists in removing stains in cleaning and is used in laundry and dish detergents.
- Bicarb is hygroscopic, meaning it absorbs moisture and hence odours.
- Finely powdered bicarb is injected into industrial exhaust stacks to de-sulphurise flue gas before it can escape into the atmosphere.
- Mix it with a bit of water to make a paste, and it relieves insect bites on your skin.
- Calms indigestion and heartburn.
- It’s the abrasive in toothpaste (also helps to remove odours in your breath).
How can we help you?
In 2022, despite the unprecedented supply chain hurdles of a global pandemic, Redox successfully distributed Sodium Bicarbonate across Australia and New Zealand to meet growing demands in both countries and Malaysia.
Redox’s Sodium Bicarbonate is available in various packing sizes, including 25kg bags and bulker bags, coming in a range of food, feed and industrial grades and conforming to FAMI-QS and Food Safety Regulation.
Contact one of our experts to discover how Redox can be essential to your sourcing strategy.
Exciting news from one of our partners, Tessara, as they have officially launched two new products at Asian Fruit Logistica Bangkok. Two new sulphur pad products for table grapes, called Uvays Vivo and Berrisys, for the blueberry industry which extends the shelf life of blueberries during storage and transportation.
Read more below about this exciting launch.
Tessara manufactures sulphur dioxide laminated sheets (sulphur pads); the company specialises in fresh science by providing post-harvest solutions that maximise the shelf life of valuable fresh produce to minimise food waste.

“Our sulphur dioxide laminated sheets are scientifically designed for individual crops: table grapes, blueberries, raspberries, blackberries and cut flowers.”
Tessara’s production facility is BRC accredited with an AA rating and operates 24/7, 5 to 7 days a week. They have approximately 200 staff, including representatives and agents in key markets in Asia, Africa, Europe, Australia, and North and South America. Tessara offers an innovative range of products that increases the shelf life during post-harvest storage and transportation.

“…Our focus is about keeping fresh produce fresher for longer.”
“Our sulphur dioxide laminated sheets are scientifically designed for individual crops: table grapes, blueberries, raspberries, blackberries and cut flowers. All our products are for post-harvest application; they sterilise the fruit and destroy fungal pathogens, mainly Botrytis cinerea, a common cause of post-harvest rot during storage. Our focus is about keeping fresh produce fresher for longer.”
Tessara’s range of products includes UVASYS and UVASYS VIVO, the fully recyclable sulphur pad for table grapes. In addition, BERRISYS and BERRISYS LITE are for berries – blueberries, raspberries, blackberries, and FLORASYS– for cut flowers.
“A smart change in your post-harvest system can significantly affect your valuable crop’s shelf life. We enjoy discussing everything post-harvest to maximise shelf life.
Both products are available and used in South Africa and the UK, with Turkey and Georgia following suit. In Australia, the APVMA registration for Berrisys is pending for December 2022. We eagerly await this approval to assist the Australian industry with its exports and domestic market opportunities for APVMA registration. Stay tuned and contact us if you have any questions regarding this exciting new product.
Did you know that Hydrochloric Acid is also known as HCL, muriatic acid, or spirits of salt? It’s utilised in a variety of industrial and commercial settings. For those who work in industries that use this chemical, it’s vital to understand the most common applications, what they accomplish, and what you need to know to handle them safely and responsibly.
What Is Hydrochloric Acid?
Hydrochloric Acid is an odourless, colourless solution of hydrogen chloride in water with a pungent smell. But behind this almost invisible veneer lies a powerful punch. For instance, Hydrochloric Acid can react with metals to form an explosive gas. Yet, it can also be found in many home cleaning products.

1150 kg IBCs housed in our warehouse, classified Hazard Class 8, are for corrosive materials, defined as substances that can cause significant harm to living tissue and/or corrode steel and aluminium if they leak.
Hydrochloric Acid is classified as a class 8 hazardous product, i.e. it’s a corrosive substance and can cause burns and irritation to the skin. Due to its corrosive properties, extreme care must be taken when handling this product. Make sure to wear appropriate safety equipment when handling hydrochloric acid. Ensure to avoid direct eye contact; if this occurs, seek immediate medical advice.
We recommend you consult the safety data sheet when using, storing or handling the product.
Hydrochloric Acid in the market and its many uses
This potent acid is found in many industries and has a wide range of uses.
What is Hydrochloric Acid used for?
The most significant end uses for Hydrochloric Acid are cleaning, the productions of fertilisers and dyes, steel pickling, oil well acidising, food manufacturing, producing calcium chloride, and ore processing.
How it’s used also varies significantly. For instance, in water treatment, it’s used to control pH levels, or in swimming pools, it can help remove any stubborn algae from the floors and walls of your pool. In acidising oil wells, it helps remove carbonate reservoirs, or limestones and dolomites, from the rock. It’s used in laboratories for acid-base titrations and for producing organic and inorganic compounds like PVC.
We also find substantial use of Hydrochloric Acid across many other industries like:
- Mining,
- water treatment,
- oil/gas,
- detergents,
- leather, building,
- textile,
- rubber,
- photography.
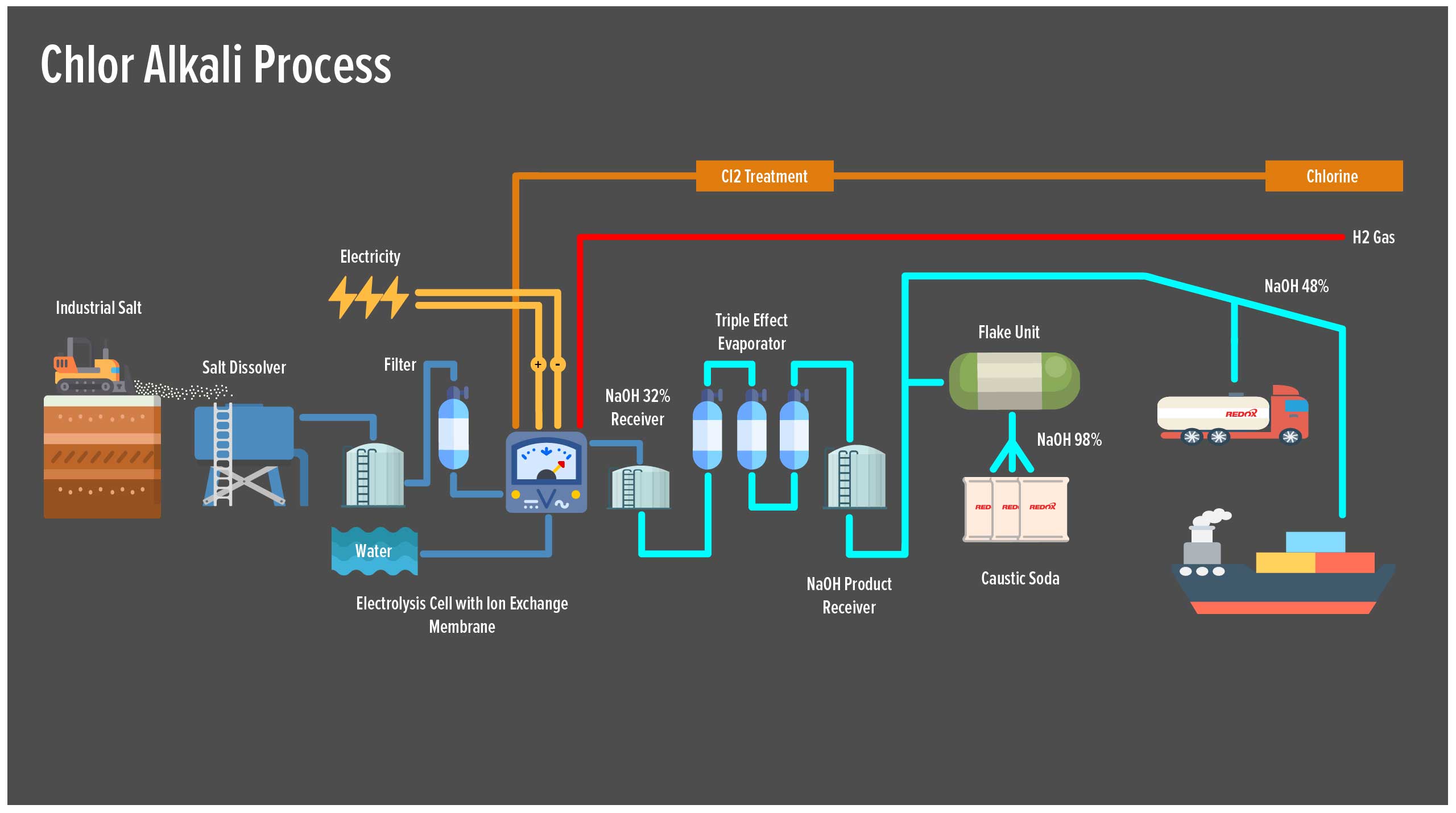
Hydrochloric acid is manufactured predominantly in industrial chlor-alkali plants around the world. The process involves the electrolysis of sodium chloride (salt) solution . This produces chlorine gas , sodium hydroxide and hydrogen gas. The hydrogen is then used to produce hydrochloric acid and ammonia.
In 2020 the global Hydrochloric Acid market size was US $7.8 billion and was expected to record a revenue CAGR of 1.5% over the forecast period through 2028.

HCL prices are usually fairly stable and tend to increase yearly as a result of CPI increases, usually as a result of the cost of production (labour /electricity costs etc). However, the imported cost of the product is highly dependent on exchange rate variations, packaging and sea freight costs. Finally the overall economics of supply and demand would also play a role in the change in the price of HCL, which is also dependent on Chlorine and liquid Caustic Soda demand, which are all part of the chlo-alkali process.
Hydrochloric Acid In The Home
In the home, you’re most likely to find it used in cleaning agents like toilet and tile cleaners, as it removes grime without reacting to many bathroom surfaces.
Is Hydrochloric Acid Corrosive?
In short, yes. Hydrochloric Acid is corrosive to organic tissues and will corrode mucous membranes, eyes, skin. It can also be corrosive to almost all metals.
How can we help you?
Redox’s Hydrochloric Acid is available in various pack sizes, including 20-litre carboys, 240 kg drums, 1150 kg IBCs and bulk tanker/Isotainers loads. The product comes in a range of strengths ranging from 6% to 33%, with 32% hydrochloric acid being the main commonly used strength.
Contact one of our experts to discover how Redox can be essential to your sourcing strategy.
Dextrose is the name of a simple sugar mainly derived from grains such as corn or root plant. Find out how Redox can become integral to your Dextrose sourcing strategy.
What is Dextrose & how is it used?
Dextrose is used in many products because it has several beneficial effects and is widely available. Its often used as a sweetener or preservative or to neutralise food that is too spicy or salty. It has a high glycaemic index, increasing blood sugar levels quickly and is an excellent source of energy.
It’s frequently used and commonly found in the below food items:
- Sauces
- Seasoning mix
- Chips
- Cured meats
- Canned foods
- Pickles
- Crackers
- Fruit jams

Dextrose added to cured or processed meat to counteract intense saltiness of cured meat and as a medium (food) for the microbial fermentation process used to reduce the pH of dry and semi-dry sausages.
Our Dextrose can also be used in animal feed /pet food and various industrial usage; however, they are more commonly used in confectionery, beverages, dry mixes and meat treatment because it adjusts sweetness and refreshes taste.
Does it have medicinal applications?
Dextrose is periodically prescribed by doctors alone or combined with other medications. It’s usually administered either intravenously or orally. It can be used to treat low blood sugar and dehydration, as well as to provide nutrition.
How can we help?
Our Dextrose is a non-GM product, available in both Monohydrate and Anhydrous variants and meets rigorous standards while retaining its authentic essence and taste. It is available in 15-25kgs bags and 800kgs bulk bags, which we routinely supply to our customers around the globe.
Redox is in a great position to meet and surpass your expectations. So contact one of our specialists by filling in the below form and have one of our team get in touch with you.
Lactic Acid is a versatile material found in a variety of innovative products that was first discovered by the Swedish chemist Carl Wilhelm Scheele in 1780 and produced commercially by Charles E. Avery in 1881.
It’s Applications in Industry?
Lactic Acid is a naturally occurring organic acid utilised in various industries, such as cosmetics, pharmaceuticals, chemicals, food, and, most recently, medical industries.
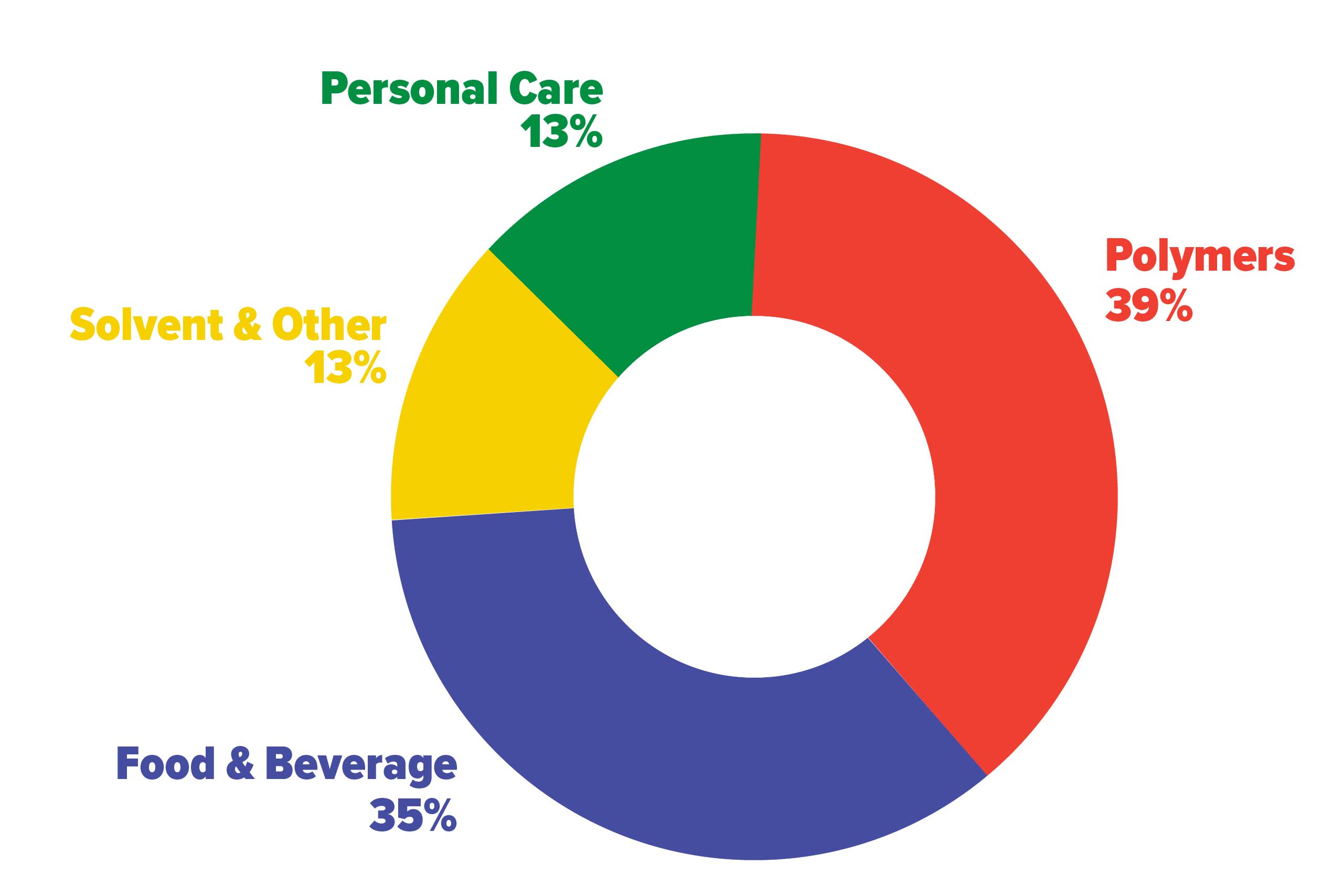
In the food industry, which accounts for a large portion of the demand (35%), it has several uses:
- It acts as an acidifier for bread,
- prevents the development of harmful bacteria in pickles, relishes, salad dressings,
- adds a mild-tasting sourness to beverages and candy,
- In dairy products, it is used to prevent fermentation and,
- It is an essential ingredient in fermented foods, like yoghurt, butter, and canned vegetables.
However, it is also used as an antimicrobial in cleaning products, has applications in the leather tanning industry, in descaling processes, in the textile industry as a mordant (fixative) for dyeing, and can be converted to ethanol, propylene glycol, and acrylic polymers in the chemical industry.
Although it’s been commercially available for a long time, it is only in recent decades that new uses have resulted in a tremendous increase in demand.
For example, its an essential building block in producing a range of new and innovative bioplastics, PLA or Polylactic Acid – the new generation of biodegradable polymers.
The use of Lactic Acid in manufacturing environmentally friendly, green solvents is another area for significant potential growth. Using it as a green solvent enriches the diversity and versatility of bio-based green solvents and could offer an effective means for designing environmentally benign synthetic systems.
Its application and innovative usefulness seem ever-growing, and in 2010, it was included in a report issued by the U.S. Department of Energy on chemicals that are considered potential building blocks for the future.
Lactic Acid from Redox
Redox is proud to work with leading manufacturers of Lactic Acid and Lactates globally and can create solutions for all customer requirements.
Redox supplies Lactic Acid in the Australian, New Zealand, Malaysian, and North American markets and is available in various packing sizes. These include 25kg carboys, 250kg steel drums and 1200kg IBCs.
We offer it in a range of varied strengths, with a powder form also available for specific applications. Our Lactic Acid conforms to the Food Chemicals Codex (FCC), ensuring our product’s overall safety and integrity.
Contact one of our experts to discover how Redox can be an essential element of your sourcing strategy.
In 1814 a German scientist named Friedrich Ferdinand Runge developed a method to extract pure cocoa powder into its most potent form. The result was a white powder containing both fat molecules (known as acids) and sugars, which create energy when digested in our body. Today that substance (caffeine) is easily extracted and used to make various products we consume daily.
Caffeine and its numerous uses
We all know that caffeine is found in our daily coffees and teas; some might also be familiar with its move into the energy drinks and the sports performance markets. The reason it has been so effective and popular in these products is that caffeine keeps us awake, alert, and active whenever a little more energy is needed.
A lesser but more recent use case is caffeine in hair care products. Caffeine boosts hair growth by stimulating the hair roots. Haircare products with caffeine also strengthen the hair itself and make it more manageable on a day to day basis.
And it’s not just used in women’s hair products. Many men face early hair loss due to the effects of the male hormone dihydrotestosterone (DHT), which acts on susceptible hair follicles. Caffeine penetrates the hair roots and stimulates them, delaying or subverting male pattern baldness.
Whether it’s brewed from an espresso, downed in sweet tea, savoured in chocolate or consumed in cola, caffeine is a mild stimulant to the central nervous system that has become a standard fixture in everyday life.

The main benefit of caffeine is that it can help promote hair growth straight from the root in the initial phases of hair growth
Synthetic v Natural Caffeine
These days the majority of the caffeine used in the market is synthetically derived as it is more cost effective to produce than the natural manufacturing process. Caffeine is found naturally in plants that humans have been consuming for thousands of years such as fruit, leaves, coffee beans, cacao, and guarana plants.
Caffeine: did you know?
Here are some fascinating facts about caffeine to consider:
- Coffee, soft drinks, and tea account for 97% of beverage caffeine consumed in the United States, with coffee accounting for the most significant proportion.
- Studies suggest that it can improve mood and concentration, sharpen your focus, and increase lifespan.
- Caffeine has been shown to enhance physical performance by 3%. Aside from telling your brain it’s not tired, caffeine improves calcium release in our muscles, allowing them to work harder.
How can we help?
Working with a supplier that can guarantee consistent quality is critical; Redox is in a great position to meet and exceed your expectations.
We offer both synthetic and naturally derived sources of caffeine across all regions. The supply of this item has been challenging for the past couple of years and requires a lot of planning to ensure continuity of supply. To avoid disruptions, Redox encourages its customers to plan ahead assuming long lead times than in the past.
Contact one of our specialists to learn how Redox can be an integral part of your sourcing strategy.
Benzoic acid was famously observed in the 1500’s by a well-known French astrologer, physician, and reputed seer, this naturally occurring aromatic carboxylic acid has an equally historic connection to Redox.
The sixteenth century saw the discovery of benzoic acid with Nostradamus (1556) first describing the dry distillation of gum benzoin, followed by Alexius Pedemontanus (1560) and Blaise de Vigenère (1596), but it was Justus von Liebig and Friedrich Wöhler determined the chemical structure.

To produce benzoic acid, Nostradame dry-distilled gum benzoin, the balsam resin from trees of the genus Styrax.
Benzoic acid has a long history here at Redox also and has remained in our product line for 56 years. Our founder, Roland Coneliano, dealt with our current principal from the outset and found success in the coatings and resins sectors early on; a customer we still supply to today.
What Is Benzoic Acid?
Benzoic Acid is a white or colourless compound with the formula C6H5COOH. Benzoic Acid occurs naturally in a range of plants, where it serves as an intermediary in the biosynthesis of metabolites. It is the simplest aromatic carboxylic acid. Originally, it was only obtainable from gum benzoin, a balsamic resin found from a range of plants/trees. Most commonly, it’s used as a preservative for a range of foods, as it extends shelf life without effecting taste or smell.
What is Benzoic Acid used for?
Benzoic acid is produced in large quantities and utilised for a variety of reasons across a diverse range of industries, including the production of perfumes, dyes, topical medicines, and insect repellents. Other uses are:
- As a preservative in foods as it can fight off fungi, bacteria and assist in extending shelf life, it’s also ideal for preserving acidic foods, jams, pickles, fruit juices, aerated drinks and it doesn’t affect taste or odour.
- To stop corrosion when used in emulsion formulations and used in alkyd resins.
- In animal feed it regulates the gut, assists in digestion, nutrient absorption and helps the animal feel fuller.
- Cosmetics, where it acts as a soothing agent and in combination with salicylic acid, it can ease irritation with eczema, insect bites and fungal infection.
How is Benzoic Acid Manufactured?
Benzoic acid is a natural component of numerous plants, animals, and berries (cranberries, plums, strawberries, apples, and prunes). As a result, it can also be found in many food products, including milk products such as yoghurt.
Synthetic manufacturing of benzoic acid occurs in the chemical reaction between Toluene and oxygen at high temperatures with a catalyst of cobalt and Manganese salts.

Benzoic acid can be used as a preservative given that it increases storage stability and mitigates corrosion when used in emulsion formulations, liquid detergents, paints, polishes and waxes.
Redox supplies benzoic acid across a wide range of industries, including coatings, construction, cosmetic, food and animal feed. We have also made significant inroads into the multinational beverage business with our FCC USP grade of Benzoic.
We have benzoic acid available in Australia and recently introduction sales to the US market. Redox can supply 25kg plastic vacuum sealed bag and 500kg TYPE C antistatic bags specifically for the coatings industry.
Contact us today and speak to one of our industry specialists to find out more.
Our Partnering Manufacturers