Yeast supplements include live yeast, yeast cell wall (YCW), purified outer layer of cell wall components such as mannooligosaccharide (MOS) and β-glucans, purified inner layer of cell wall, and yeast extract products. It is generally accepted that yeast products can improve pig gut health via improved gut integrity and maintenance of a healthy microbiome, leading to increased nutrition absorption, and thus improved overall pig performance.
Live yeast refers to yeast cells that are still alive and active. It is often used to improve digestion and promote gut health for ruminant animals.
After autolysis (using the yeast’s own enzymes) or hydrolysis (using added exogenous enzymes), the yeast extract and cell walls are separated using centrifugation before being dried. Usually, the yeast extract via autolysis contains higher levels of yeast nucleotides. However, the yeast extract via hydrolysis is more applicable due to a low salt concentration.
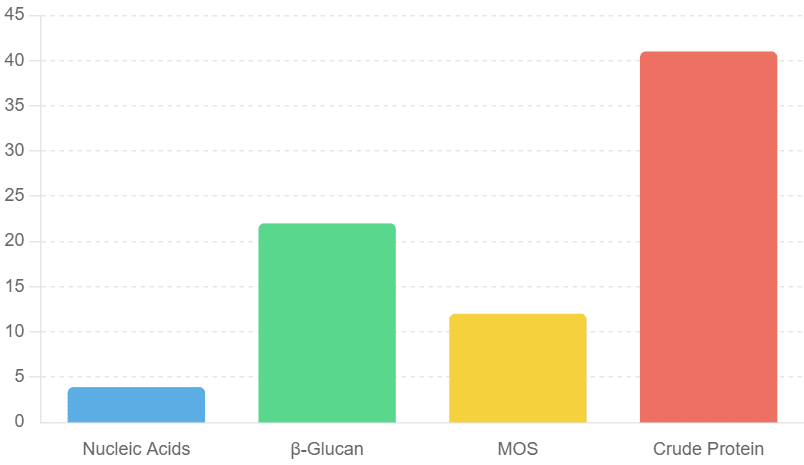
The composition of autolyzed yeast (AY)
The composition of autolyzed yeast (AY) has been summarized as 3.5-3.9% nucleic acids, 11-22% of β-glucan, 3-12% MOS, and 30-41% crude protein. Adding 0.2-0.5% AY to pig diets could improve the performance and immune function of weaned and finished pigs.
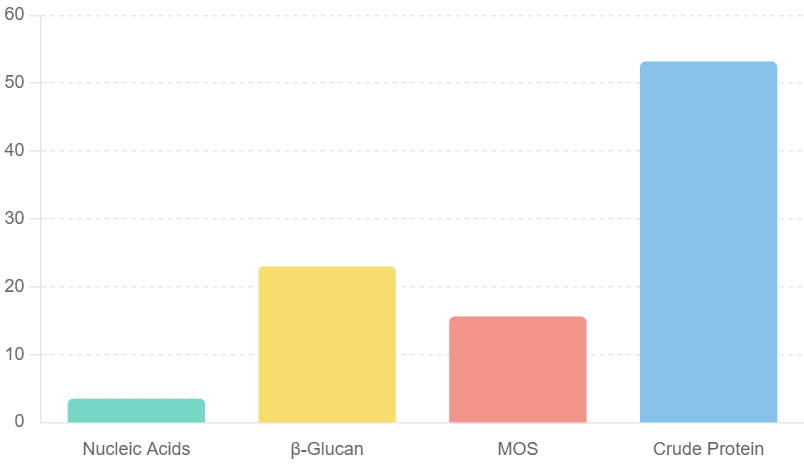
The composition of hydrolysed yeast (HY)
The composition of hydrolysed yeast (HY) includes 3.5% nucleic acids, 22.43-23% β-glucan, 15-15.6% MOS, and 40-53.2% crude protein. The effective inclusion rates of HY in diets for the weaned, grower, and finisher periods are 0.1-0.2%, 0.05-1.0%, and 0.1%, respectively.
The yeast cell wall fraction is mainly composed of polysaccharides, making up 15 to 30% of the dry weight of the whole cell. The inner layer of the cell wall mainly contains β-D-glucans which is used as the organic mycotoxin binder. Supplemental levels of the mycotoxin binder are recommended around 0.1 to 0.2%
The outer layer of the yeast cell wall is made of MOS and β-glucans. Adding purified β-glucan to early weaned piglets dets showed protective effects against E Coli infection by reducing bacterial excretion and diarrhoea. The recommended dosage of β-glucan is 100-200 ppm or 100 -200 g/MT feed. The purified MOS can attach the bacterial surface, and then protect the colonization of bacteria in the intestinal tract. Supplementation of MOS in the range of 500 to 1000 ppm or 500 g to 1 kg /MT feed.
In addition, approximately 1-20% of total nitrogen in yeast are derived from nucleic acids. Nucleotides are significantly required by cell replication process, particularly intestinal epithelial and lymphoid cells. Nucleotides are also important in immune system maintenance and prevent oxidative stress. The purified Nucleotide inosine monophosphate (IMP) is commercially available and recommended supplemental levels are ranged 500 ppm to 1000 ppm or 500 to 1000 g/Kg feed.
A study compiled by our Redox Animal Nutritionists.
Yucca schidigera is an herbaceous plant of lily family, native to the deserts of the southwestern United State and northern Mexico. It contains two active compounds: at least 10.5% steroidal saponins and polyphenols. Saponins are similarly in structure to Glucocorticoids in animals.
Previously It was thought that the Yucca extract could bind ammonia and therefore reduce its emission from pets and other animals. However, ammonia concentrations in the gut are at least 100-fold greater than the concentrations of the yucca extract. Currently it is believed that the high circulating levels of Glucocorticoid in response to various oxidative stress including heat stress result in enhanced degradation of amino acids to generate ammonia and reduced protein synthesis in skeleton muscle.
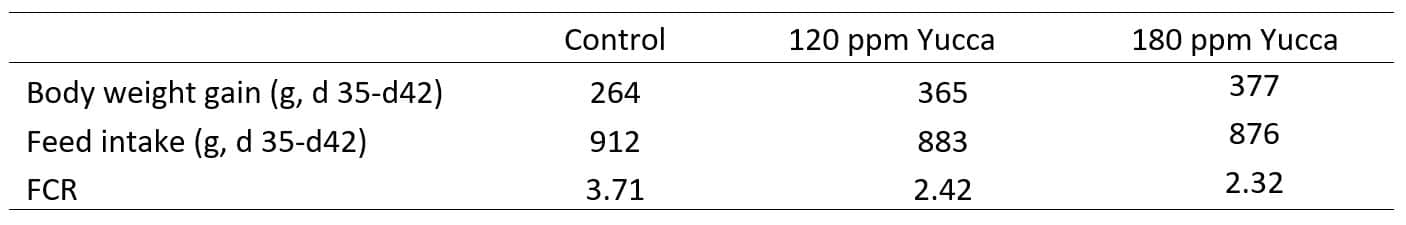
Under conditions of the oxidative stress including heat stress, the Yucca extract can block the bonding of glucocorticoids to their receptors in cells. Thus, the yucca extract could alleviate the oxidative stress including heat stress and reduce ammonium emission in the stress situation (Table 1). When broiler chickens were housed at an average ambient temperature of 24˚C, dietary supplementation of Yucca extract (120 ppm and 180 ppm) did not affect the feed intake or body weight gain (Wu, 2018). However, in table 1, when the ambient temperature was naturally elevated from 27˚C to 37˚C, the Yucca supplementation improved body weight gain by 38-43% and FCR 46% to 52% compared with the control group.
Table 1 The effect of the addition of the Yucca extract on broiler chicken performance under the heat stress
Yucca saponins also have anti-protozoal activity by supressing protozoal infection of the intestine. It is reported that Yucca saponins could bind with cholesterol in protozoal cell membrane and destroy the integrity of the protozoal membrane. Coccidiosis is one of the main challenges for the worldwide poultry industry. Although coccidiostats are often rotated, the poultry industry is challenged by resistance of the Eimeria parasites to these treatments. In a recent trial conducted in the Europe, Kozlowski et al (2022) reported that both the anticoccidial and 500 g/Mt Yucca supplementation showed significant improvement in body weight (2150 g and 2058 g vs. 1998 g, Table 2)
It is now also reported that Yucca polyphenolics have anti-inflammatory activity and could prevent arthritis. The generation of reactive oxygen species such as nitric oxide is an important factor in the development of rheumatic arthritis. Yucca polyphenolics may inhibit nitric oxide production and prevent arthritis.
Table 2. The effect of supplemental Yucca extract on challenged chicken performance (d1-35)
A study compiled by our Redox Animal Nutritionists.
It is well known that in the current floor pen with deep litter system, day old broiler chickens could benefit from non-starch polysaccharides components in the litter to establish the dynamic microbiota. However, when the intestinal microbiota becomes complex and diversified over time and environmental conditions, long term exposure to faeces and ammonia pollution environments has a higher risk of infection with pathogenic bacteria and parasites.
For decades, chicken enteric disease including necrotic enteritis (NE) and coccidiosis have been controlled by using antibiotic growth promoters (AGP) and coccidiostats. However, due to the development of antibiotic resistance in bacteria and coccidiosis, which is a threat to animal and human health, the use of AGP and coccidiostats has been restricted or banned in the poultry industry. Thus, alternatives production strategies including vaccinations, organic acids, prebiotics, probiotics, essential oils, postbiotics or yeast peptides to control NE and coccidiosis have been explored.
The Glucose Oxidase (GOD) is exogenously produced by specific fungi fermentation to oxidize β-D-Glucose into gluconic acid and hydrogen peroxide, consuming large amounts of oxygen at the same time in the chicken gut. Therefore, it can protect against oxidative stress and directly kill some pathogenic bacteria or Eimeria parasites. Gluconic acid is a kind of organic acid, which acts as an acidifier in the intestine to produce the short chain fatty acids such as butyric acid. GOD also plays an important role in colour development, flavour, texture, and increasing the shelf life of food products. Due to its characteristics of producing natural acid, deoxygenation and sterilization, GOD has been defined in AAFCO list as 70.3 and widely used in animal production.
Recently, the University of New England (UNE) conducted a NE challenging trial to investigate the GOD potentials to ameliorate the impact of NE on chicken performance and intestinal health. The standard, positive controls (PC) were formulated based on wheat-sorghum-soybean meal as the commercial diets. Negative control (NC) chickens were challenged with Eimeria parasites at day 9 and clostridium perfringens at day 14. Another 4 treatments consist of PC or NC diets supplemented with antibiotics, GOD 100 g/MT, 200 g/MT and 300 g/MT, respectively.
The effect of GOD on chicken performance before challenging was shown in Figure 1. It is clearly shown that adding GOD significantly improved chicken body weight gain by 9% and FCR was improved by 10 points. From the growth point of view, AGP supplementation did not show any impact on chicken body weight gain and FCR.
However, during the challenging period (Figure 2), AGP significantly improved the chicken body weight gain and FCR compared with the positive control. Interestingly, during the recovery period (Figure 3) or chickens had the longer-term exposure to faeces and ammonia pollution, chickens in positive control and supplemented with the lowest GOD showed worst performance, but chickens in the negative control showed the best performance, possibly due to that Eimeria challenged birds boomed their immune system for the possible coccidiosis infection. It might also imply that supplementation of GOD could replace both coccidiostat AGP.
However, the lower level of GOD might have the negative effect of Eimeria vaccination.
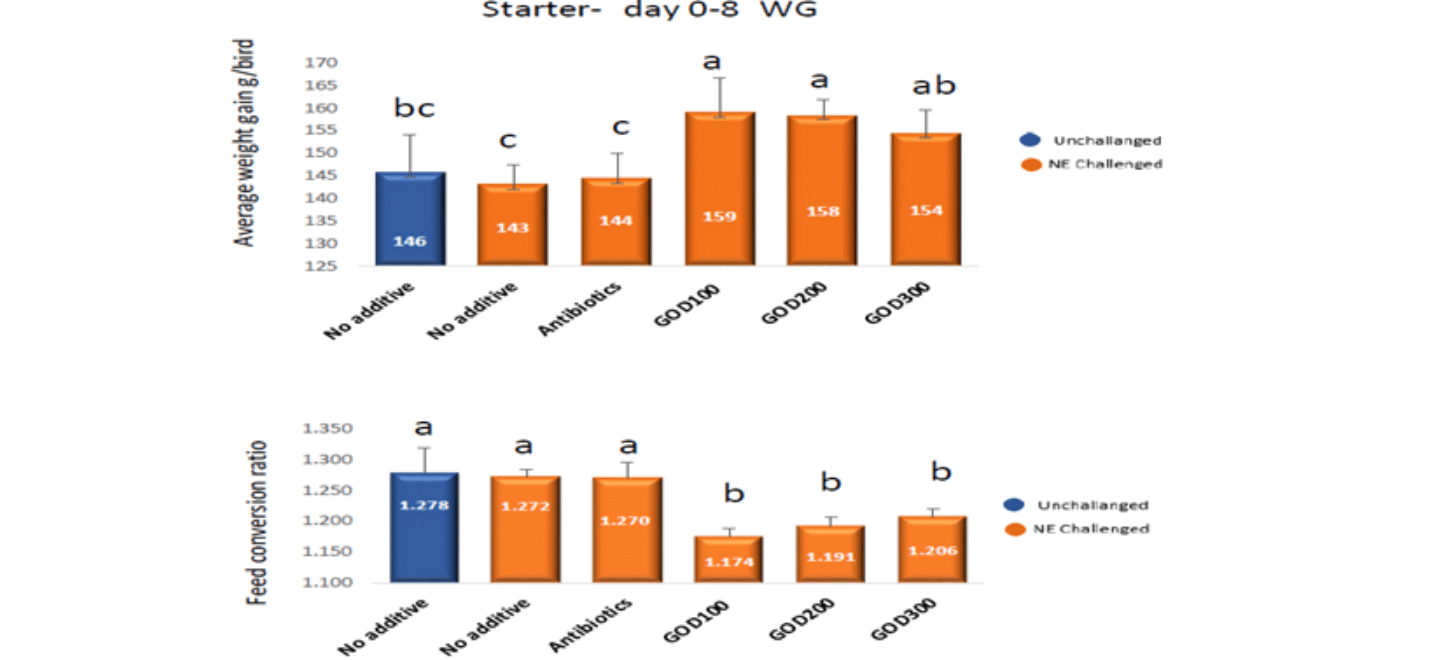
Figure 1. The effect of GOD on chicken performance before challenging.
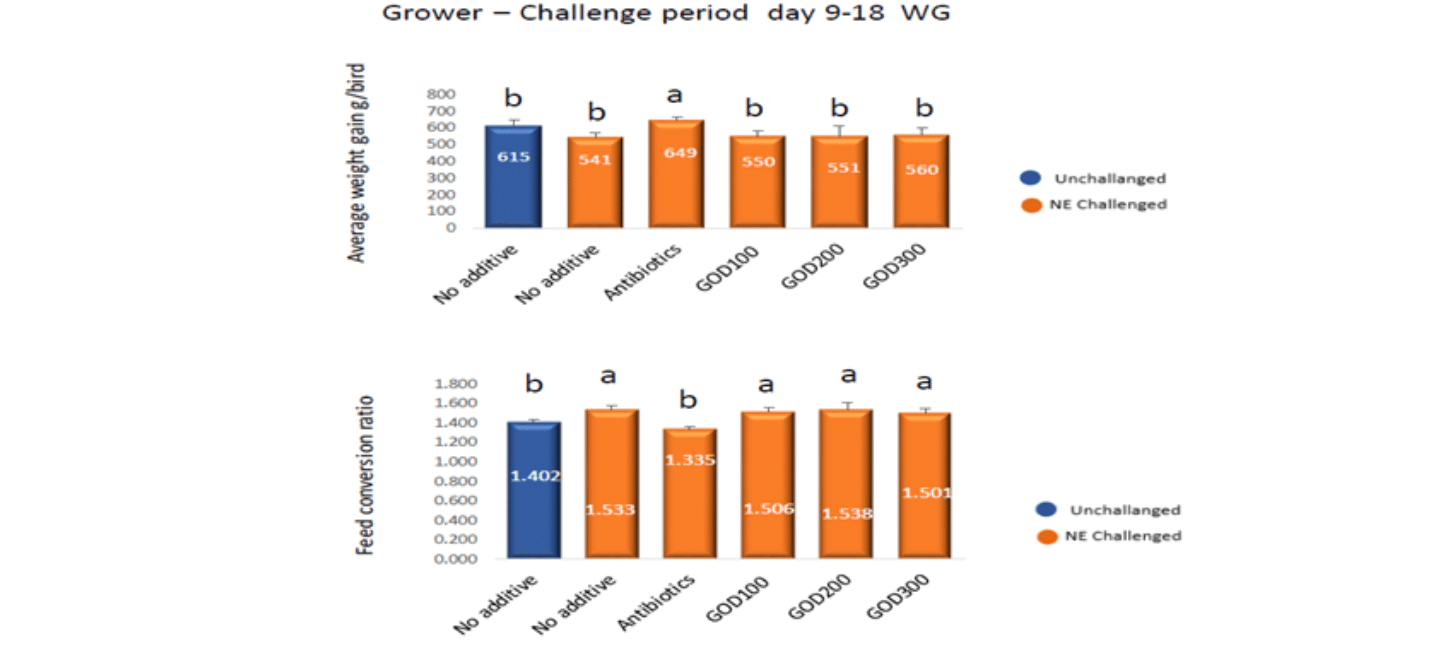
Figure 2. The effect of GOD on chicken performance during challenging.
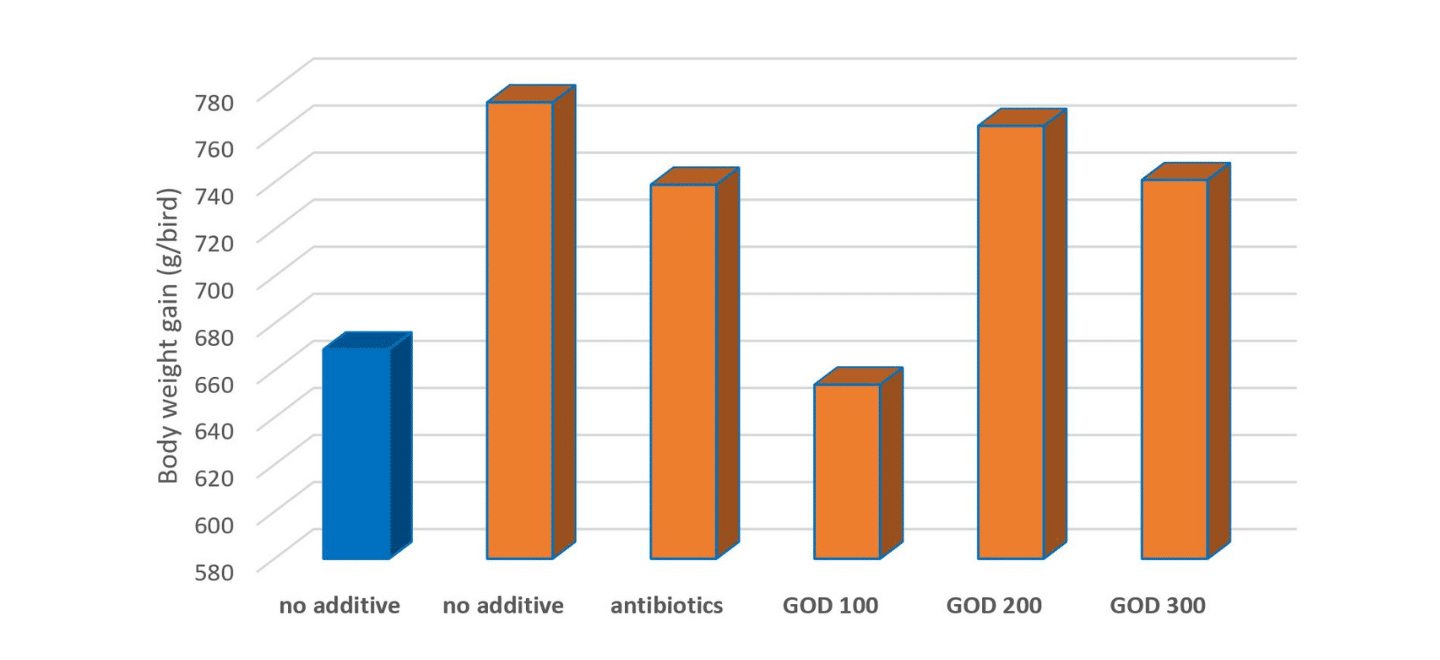
Figure 3. Efeect of GOD on chicken body weight gain after challenging period (d 28-35)
A study compiled by our Redox Animal Nutritionists.
Rumen microorganisms can utilize non-protein nitrogen (NPN) such as ammonia to synthesis rumen microbial proteins for cattle and sheep. Urea is a cheap source of NPN but the hydrolysis rate of urea in the rumen is speedy and exceeds the ammonia utilisation rate of rumen microorganism. Surplus ammonia is harmful to the animal and is also associated with increased methane production. Coated urea is designed to provide a controlled release of urea in the rumen, allowing for a more efficient utilisation of nitrogen by rumen microbes.
In Figure 1, it is clearly shown that compared with Menogen plus (the coated urea), normal urea is almost completely degraded within 20 minutes. On the other hand, the degradation rate of soybean meal or canola meal is too slow. Within 8 hours, the degradation rate of soybean meal and canola meal is about 50% and 30%, respectively, considered as the good source of rumen undegraded protein.
However, less ammonia concentration may not provide sufficient nitrogen source for rumen microorganisms to effectively synthesise rumen microbial protein. Therefore, addition of the coated urea could provide stable nitrogen supply for rumen microbes. It is reported that providing 90 grams coated urea per day per cow could replace 450 grams soybean meal and increase milk production by 0.85 kg per day per cow.
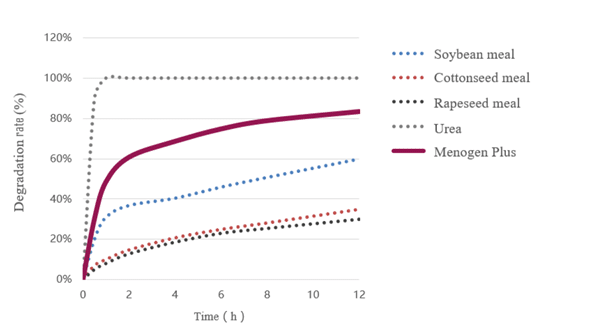
Figure 1. The degradation rate of different nitrogen sources in Rumen
In summary, this controlled release urea can lead to several benefits in terms of methane reduction and overall animal nutrition:
- Reduced ammonia levels:
The coated urea undergoes gradual hydrolysis in the rumen, releasing ammonia at a controlled rate. This controlled ammonia release ensures a stable nitrogen supply for rumen microbes, reducing excess levels. High ammonia levels in the rumen are associated with increased methane production.
- Improved microbial protein synthesis:
The coated urea supports the growth of rumen microbes by providing a steady and controlled supply of ammonia. These microbes are responsible for breaking down fibrous materials and producing microbial protein. Efficient microbial protein synthesis can lead to improved feed digestion and reduce d methane emissions.
- Optimised nitrogen utilisation:
The coated urea allows for better synchronisation between available nitrogen and microbial needs. This synchronisation can lead to improved utilisation of dietary protein and reduced excretion of nitrogen in the form of urea. Lower nitrogen excretion can contribute to reduced ammonia levels and subsequently, reduced methane production.
- Balanced rumen environment:
The coated urea helps in maintaining a stable rumen PH and stable pH conditions are conductive to the growth of specific microbes that produce less methane during feed digestion.
- Increased fibre digestibility:
Improved microbial efficiency and balanced rumen pH can enhance the digestion of fibrous materials. Enhanced fibre digestion results in fewer substrates available for methane-producing microbes, leading to reduced methane emission.
A study compiled by our Redox Animal Nutritionists.
Layer chickens’ dietary fibre comprises a significant part of plant feedstuffs and is chemically defined as the non-starch polysaccharides (NSP). The NSP include various fibre types such as lignin, β-glucans, arabinoxylans, uranic acid, galactose, and mannose.
Soluble NSP such as arabinoxylans in wheat or mannans in soybean meal will increase the chicken gut viscosity, resulting in detrimental effect on chicken performance and egg production. Therefore, adding xylanase and β-mannnase blend to layer chicken wheat-soy based diets could remove these anti-national factors and reduce the energy cost of immune responses.
Recently, White et al (2021) reported that adding β-mannnanase to the corn-soy based diets for laying hens significantly increased egg production by 6% (88.55% vs 94.94%). In laying chickens feed formulation, adding β-mannanase could save about 100 Kcal/kg apparent metabolizable energy (AME) and 1%-unit crude protein level.
In Germany, VTR conducted a layer chicken trial to investigate the effect of the exogenous xylanase on egg production and apparent N-corrected metabolizable energy (AMEn). On the base of a standard corn-wheat-soybean meal diets, 100, 150, and 200 grams/Mt xylanase were added to include a total of 4 treatments. The effect of xylanase on egg production and egg quality was listed in Table 1. It is clearly shown that adding 200 g/MT xylanase increased egg production by 1.8% and significantly reduced dirty and broken eggs. It cam also increase about 150 kcal AMEn.
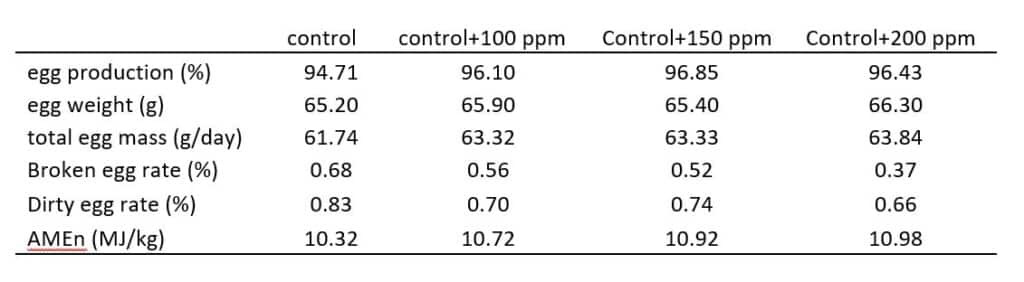
Table 1. The effect of Xylanase on egg production and egg quality (week 25 to week 32)
On the other hand, the insoluble NSP such as lignocellulos have a positive effect on animal health and productivity. In particular, for free rage layer chickens, feather pecking, and cannibalism are a serious problem and the increased insoluble NSP or fibre has been widely shown to reduce feather pecking and cannibalistic behaviours. This effect is generally attributed to increased time spent eating, thus reducing redirected behaviours. JELUVET®lignucellulose contains 67.7% crude fibre, mainly comprising of cellulose and hemicellulos (62%). Therefore, it is a good source of insoluble NSP to reduce feather pecking for free range layer chickens.
A study compiled by our Redox Animal Nutritionists.
Chicken gut health is crucial in antibiotics-free period. Some alternatives such as probiotics, postbiotics, prebiotics, bile acids, antimicrobial peptides, essential oils, and vaccinations have been gradually accepted in poultry production.
Recently, a new alternative feed enzyme, glucose oxidase (GOD), has drawn attention to poultry and swine industry.
The GOD is exogenously produced by specific fungi fermentation to oxidize β-D-Glucose into gluconic acid and hydrogen peroxide, consuming large amounts of oxygen at the same time in the chicken gut. Therefore, it can protect against oxidative stress and directly kill some pathogenic bacteria or Eimeria parasites.
Recently a thermal stable GOD is commercially available, and Table 1 clearly shows that at 85 °C, the enzyme recovery rate of this GOD maintains 92%.
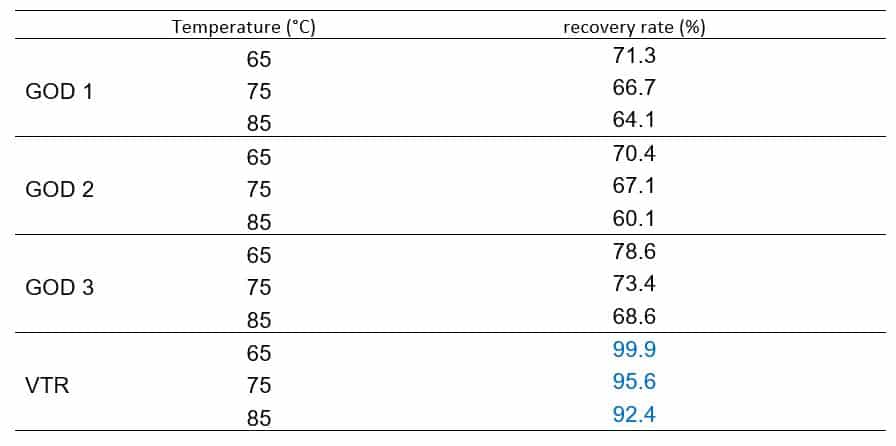
Table 1. Thermostability of different GOD (3 minutes bath incubation)
In general, adding 2000 U/kg GOD to layer chicken feed increased egg production from 88% to 90% within 4 weeks of production. Egg weight increased from 60.8 to 61.4 gram/egg.
Adding 3000 u/Kg GOD to broiler chicken diets increased body weight gain by 3.44% and FCR was improved by 6 points.
A study compiled by our Redox Animal Nutritionists.
Currently most dogs are fed highly processed food that is quite different from canine ancestral diets. Sweeteners are quite often included in processed dog food to mask unpleasant taste or enhance the acceptance of the food.
In the recent years, due to health issues, numerous low-calorie sugar free sweeteners have been developed to replace sucrose. Table 1 shows relative sweetness, acceptable daily intake (ADI) of some of some sweeteners.

Table 1 The relative sweeteners and ADI (mg/kg body weight/day) of some sweeteners
It is noteworthy that xylitol will result in fatal reduction in blood glucose levels and the liver failure has been reported in dogs.
Stevia is a sugar substitute made from the leaves of the stevia plant. it is about 250 to 300 times sweeter than sucrose. However, some people complain it is bitter.
Neohesperidine Dihydrochalcone (NHDC) is a sensory sweetener derived from the hydrogenation of Neohesperidine, a flavanone which is found in the immature fruit of bitter oranges. It is about 1000-1800 times as sweet as sucrose. However, commercially available Sugarex containing 10% NHDC is about 150 times as sweet as sucrose and its recommended dosage rates in dog dry food is 50-150 g/MT.
More importantly, all those alternative sweeteners are not effective substrates for plaque bacteria and therefore less likely result in dental caries. However, it is noticed that approximately 50-88% dogs over three years of age have periodontal disease and adding 0.6% sodium hexametaphosphate significantly reduced the area covered by dental calculus. In cats, it is reported that adding 1.2% lactic acid in a maintenance food significantly inhibited oral substrate accumulation.
Adding NHDC to weaning piglets, calf and fish diets also significantly increase the feed intake of young animals.
Report by Redox Animal Nutritionists
It is well known that reduced protein diets in broiler chickens could lessen nitrogen excretion and environmental pollution. However, the successful strategy for broiler chicken production fed on reduced protein diets largely depends on both exact amino acids balance and sufficient quantity.
Currently ideal amino acids profile and absolute standardised ileal digestible lysine concentrations provide this recommendation on amino acids balance and quantity, respectively. In reduced protein diets, if any amino acid is deficient or limiting, ingestion of excess of other amino acids will result in accumulation of these amino acids in plasma, leading to the reduction in feed intake. Figure 1 clearly shows that when the reduced protein diet is deficient in Arginine, it will result in increased plasma lysine, methionine plus cysteine, threonine, Valine, and isoleucine concentrations, indicating these surplus essential amino acids could not be degraded for muscle growth.
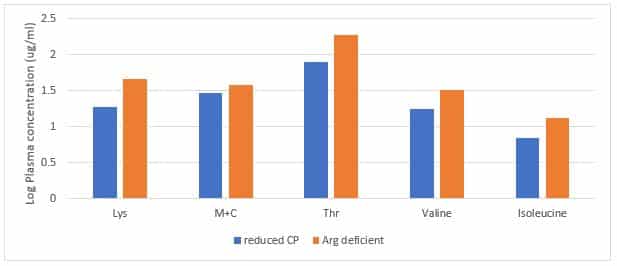
Figure 1. The effect of Arginine deficiency on plasma amino acids concentration (log ug/ml)
Arginine is one of most versatile functional amino acids because it is a precursor of several molecules, such as creatine, ornithine, nitric oxide, citrulline, proline, and polyamines. Apart from protein synthesis, it can stimulate the release of growth hormone and insulin-like growth factor 1.
Arginine also acts as a trigger of target of rapamycin (TOR) signalling system activation for reducing protein degradation. Supplementation of L-Arginine has been approved significantly increase Nitric oxide and reduce ascites related mortality.
In recent updated amino acids profile recommended by Aviagen breeder company, the SID Arginine to SID Lysine ratio has been increased significantly, up to 110% in the finisher period. A 42 days broiler chicken trial conducted in the University of Sydney clearly showed that when SID Arginine to Lysine ratio increased from 107% to 113%, plasma Lysine, methionine plus cysteine, and threonine concentrations were significantly reduced (Figure 2).
Increasing SID valine to Lysine ratio from 76% to 81%, SID isoleucine to lysine ratio from 69% to 73%, and SID Arginine to lysine ratio from 107 % to 113%, increased breast meat by 59 grams.
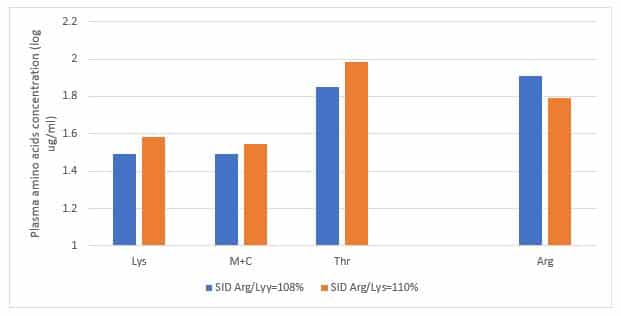
Figure 2. Plasma lysine, methionine plus cysteine, and threonine concentrations in response to dietary SID Arg/Lys ratios.
Report by Redox Animal Nutritionists
Taurine is a β-amino acid containing a sulfonate instead of a carboxylic group in α-amino acids. It is vital for organ development and aging but not for protein synthesis. Taurine is needed for membrane stabilization, has cytoprotective and cell volume regulation effects, and maintains calcium homeostasis and signalling.
Taurine deficiency impairs growth, fertility, triggers immune deficiency, muscle atrophy, and increased susceptibility of diseases, resulting in a pathological condition known as green liver in strictly marine species due to bile pigments accumulation. Under normal nutritional conditions, fish can produce taurine via methionine-cysteine metabolism pathway.
It is well known that a steady decline of fish meal inclusion levels in aqua cultural feeds has pushed higher inclusion levels of plant protein rich ingredients supplemented crystalline methionine. We assume that the higher level of dietary methionine supplementation might provide a sufficient taurine precursor to support growth rate in fish. However, Gaylord et al (2007) indicated dietary taurine supplementation was indeed beneficial fort rout fed all-plant protein diets, but supplementation of methionine above the requirement could not spare taurine.
Based on the control diet without supplemental methionine and taurine, taurine and methionine were supplemented at 0, 5, or 10 g/kg diet in a 3 x3 factorial design, respectively (figure 1).
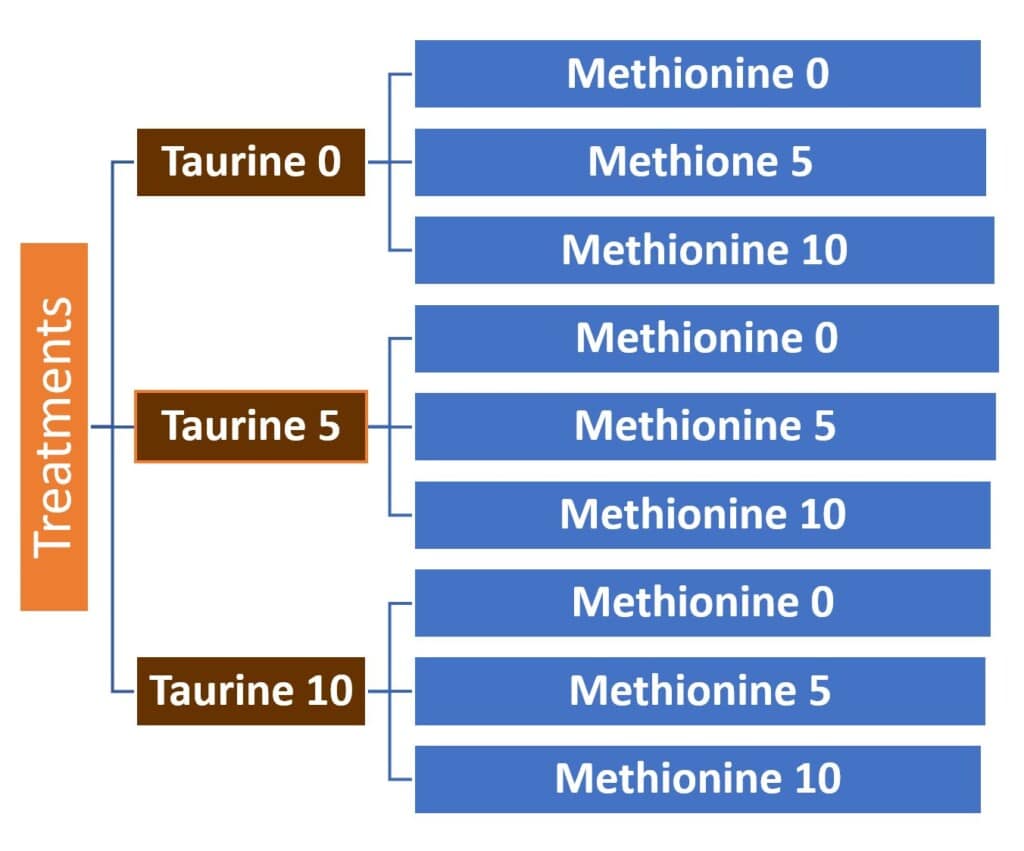
Figure 1. 9 experimental diets in 3×3 factorial design.
The relative body weight gains in 9 dietary treatments were shown in Figure 2. There was no interaction (P > 0.05) between Taurine and methionine supplementation. Adding about extra 6.5 g/kg taurine could optimize Rainbow Trout body weight gain.
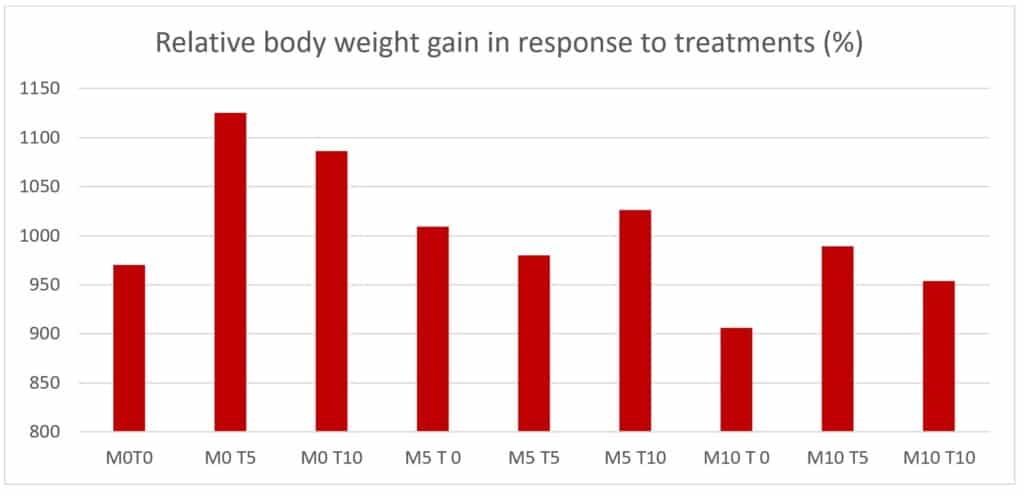
Figure 2. Relative body weight gain in response to taurine and methionine supplementation
A study compiled by our Redox Animal Nutritionists.
Gut health is important for weaning piglets, in particular when the high dosage of ZnO and antibiotics are removed from piglets feeds..
Apart from the dietary nutrients’ digestion and absorption, the gut is the largest immune organ in pigs and can be divided into four protection layers (Figure 1, Gao et al, 2020).
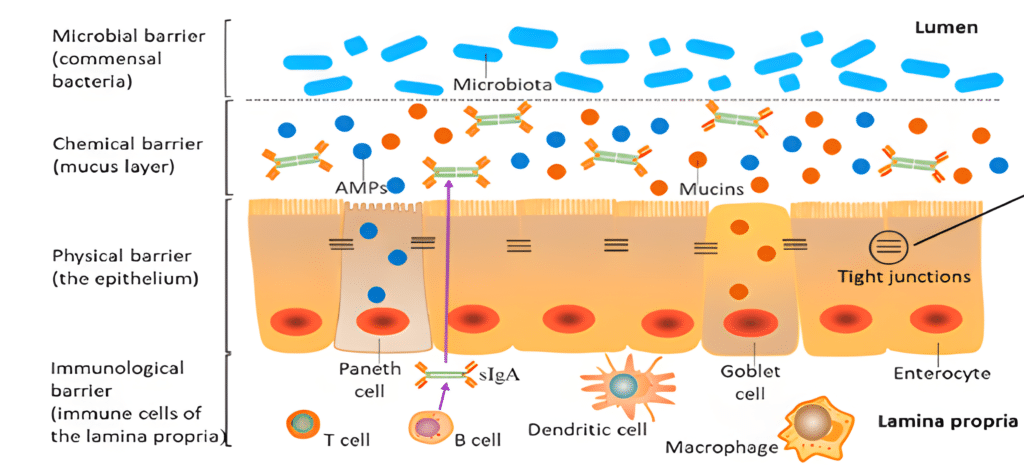
Figure 1. The different protective layers of the intestinal barrier
The commensal bacterial barrier is a complex environment playing a key role in maintaining the gut health; the chemical barrier consists of the mucus layer secreting mucins and antimicrobial peptides; the physical barrier is composed of epithelial cells and the immunological barrier harbouring the immune cells that secreted immune mediators such as cytokines and antibodies.
Traditionally bile acids are bio-surfactants, assisting intestinal digestion and absorption of lipids and fatty soluble vitamins such as Vitamin A, D, E and K, and thereby improving nutrient utilization. Recently, in the gut epithelial cell, bile acids are identified as signalling molecules to activate a couple of nuclear receptors, namely farnesoid X receptor (FXR), pregnane X receptor (PXR), Vitamin D receptor (VDR) and G-Protein coupled receptor (TGR5). Therefore, bile acids play an important role in regulating epithelial barrier functions.
In 2018, Ipharraguerre et al indicated that currently all alternatives including probiotics, prebiotics, organic acids, essential oils, antimicrobial and plants extracts to antibiotics or high dosage of ZnO underlined their growth promotion action. They demonstrated that either antibiotics or the high dosage of ZnO could activate bile acids receptors and consequently spare nutrients for growth and improve the metabolic efficiency of antibiotics or the high dosage of ZnO treated animals. Therefore, it is necessary for adding the exogenous bile acids to antibiotics free or the high dosage ZnO free diets in weaning piglets.
Based on experimental data in China, Belgium, adding 350 grams per MT feed commercially available exogenous bile acids could improve daily weight gain by 9% and FCR by 7 points.
Report by Redox Animal Nutritionists
Our Partnering Manufacturers